
Given: molecular formula and mass of sampleĪsked for: mass percentage of all elements and mass of one element in sampleĪ Use atomic masses from the periodic table to calculate the molar mass of aspartame.ī Divide the mass of each element by the molar mass of aspartame then multiply by 100 to obtain percentages.Ĭ To find the mass of an element contained in a given mass of aspartame, multiply the mass of aspartame by the mass percentage of that element, expressed as a decimal. Calculate the mass of carbon in a 1.00 g packet of Equal, assuming it is pure aspartame.Calculate the mass percentage of each element in aspartame. Chandravanshi Background This study was conducted to develop fast and cost effective methods for the determination of caffeine in green coffee beans. Caffeine Molecule Metal Wall Decor, Laser Cut Caffeine Metal Wall Art, Wall Hanging, Chemical Formula Wall Decation, For Coffee Lovers Decor (93) Sale Price 68.00 68.00 85.00 Original Price 85.00 (20 off) FREE shipping.Water activity refers to unbound water found in coffee which is not bound to coffee molecules and which is free to. (Rounding to the correct number of decimal places can, however, cause the total to be slightly different from 100%.) Thus 100.00 g of sucrose contains 42.12 g of carbon, 6.48 g of hydrogen, and 51.41 g of oxygen to two decimal places, the percent composition of sucrose is indeed 42.12% carbon, 6.48% hydrogen, and 51.41% oxygen. Molds can develop at 15 moisture and above. 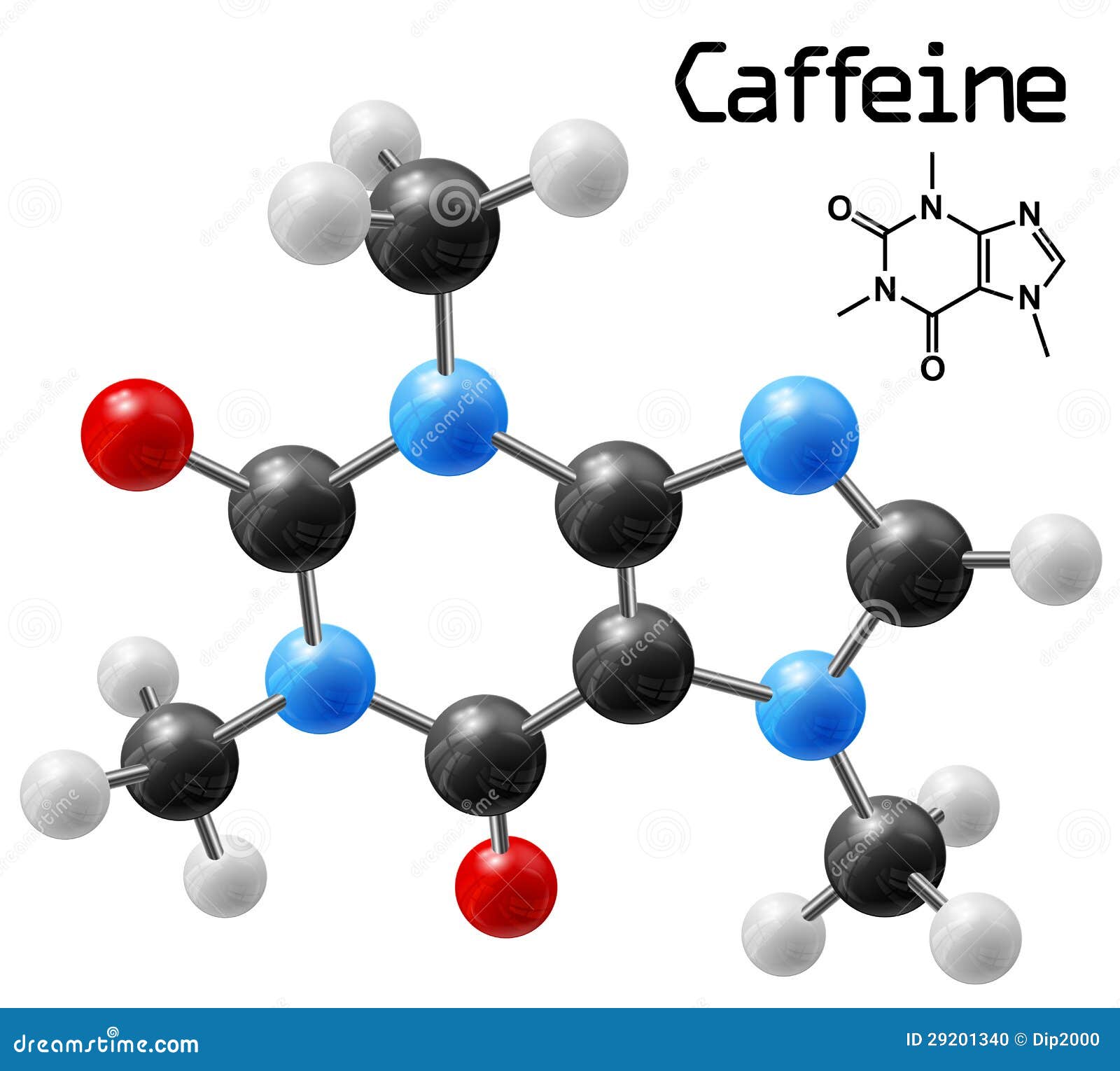 
If the sum is not 100%, you have made an error in your calculations. You can check your work by verifying that the sum of the percentages of all the elements in the compound is 100%:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
